|
The zeolite catalyst has sites which can remove a hydrogen from an alkane together with the two electrons which bound it to the carbon. It also produces high proportions of branched alkanes and aromatic hydrocarbons like benzene. The zeolites used in catalytic cracking are chosen to give high percentages of hydrocarbons with between 5 and 10 carbon atoms - particularly useful for petrol (gasoline). The alkane is brought into contact with the catalyst at a temperature of about 500 ☌ and moderately low pressures. You may have come across a zeolite if you know about ion exchange resins used in water softeners. They are, of course, associated with positive ions such as sodium ions. These are complex aluminosilicates, and are large lattices of aluminium, silicon and oxygen atoms carrying a negative charge. Modern cracking uses zeolites as the catalyst. 
The octane is one of the molecules found in petrol (gasoline). The ethene and propene are important materials for making plastics or producing other organic chemicals.  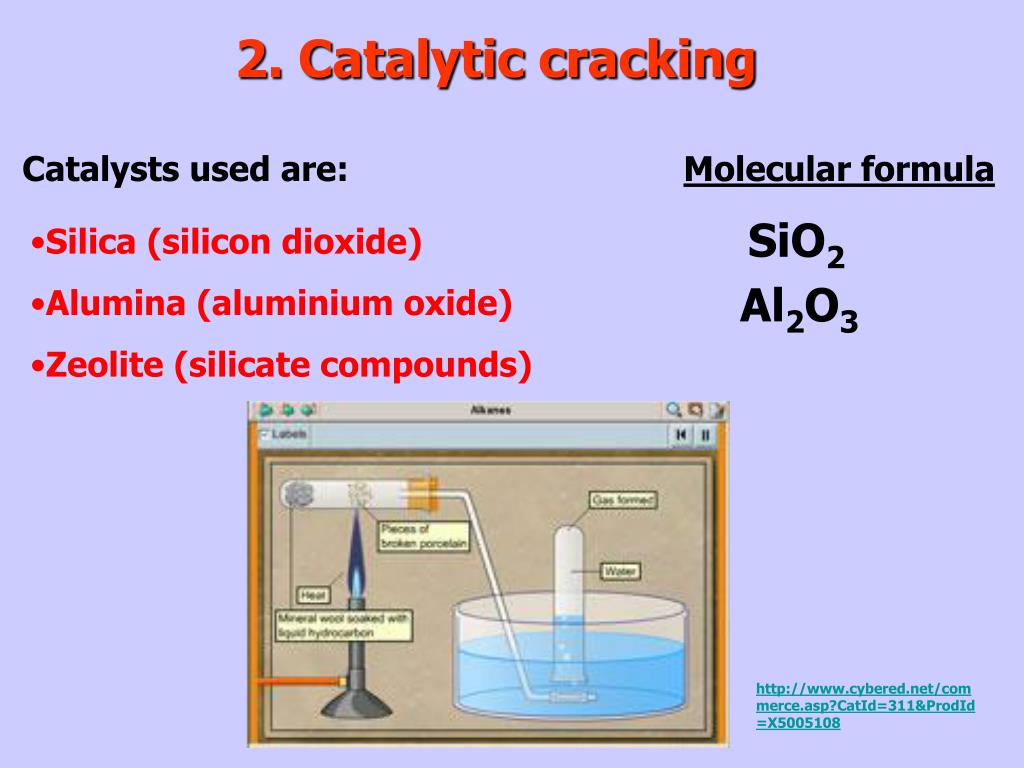
This is only one way in which this particular molecule might break up. Or, showing more clearly what happens to the various atoms and bonds:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
